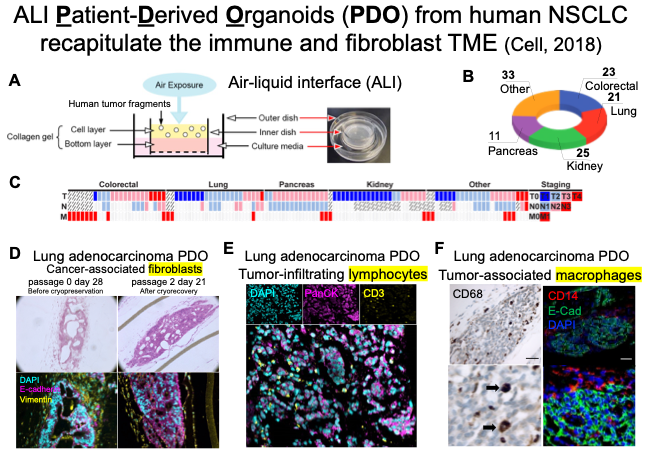
Project 1 is translational and uses clinical specimens and patient-derived models to test the hypothesis that tumor macrophages and tumor fibroblasts promote acquired resistance via paracrine signaling interactions sensed by cancer cells and converging on survival pathways.
Aim 1:
Define the roles of cancer cell/macrophage networks in promoting acquired resistance to molecular therapies in EGFR- and KRAS-driven NSCLC.
Aim 2:
Define the role of cancer cell/fibroblast signaling networks in promoting acquired resistance to molecular therapies in EGFR- and KRAS-driven NSCLC.
(For additional information, please visit NIH RePORTER)
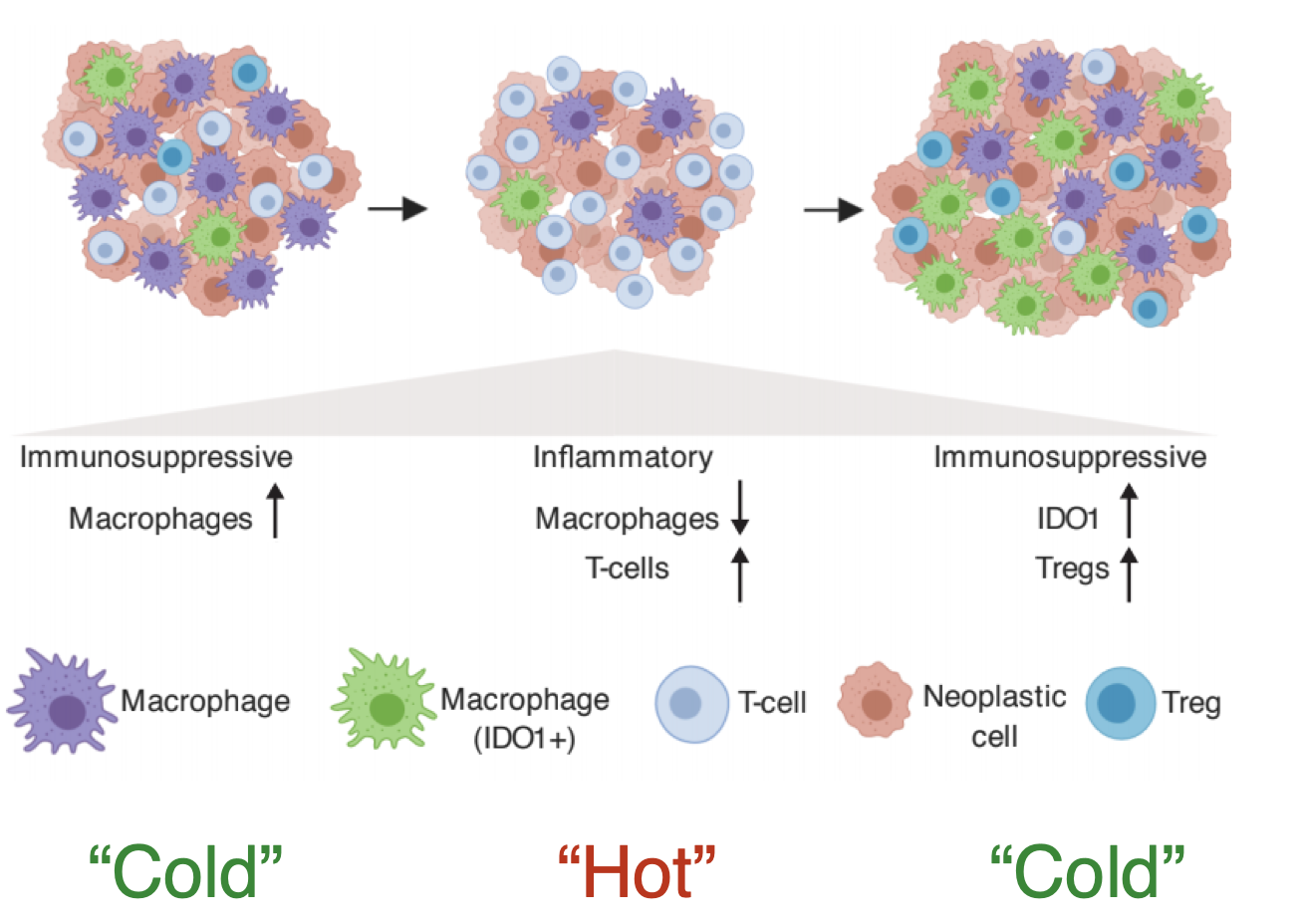
With the hypothesis that the molecular subtypes of EGFR and KRAS mutant NSCLC tumors with acquired mutation targeted drug resistance are associated with an immunosuppressive TME including both immune and non-immune stromal cells that can be therapeutically targeted in a way that will enhance the efficacy of drugs targeted to intrinsic resistance pathways, project 2 has two specific aims:
Aim 1:
Develop and characterize existing and new models of mutant EGFR and KRAS inhibitor acquired resistance including PDXs and PDX-derived organoids and characterize mechanistic relationships between molecular subtypes and the TME, including immune and non-immune stroma.
Aim 2:
Define treatment strategies in mutant EGFR and KRAS inhibitor acquired resistance models based on targeting pathways implicated in current and future studies of acquired resistance caused by tumor-TME cell networks.
(For additional information, please visit NIH RePORTER)
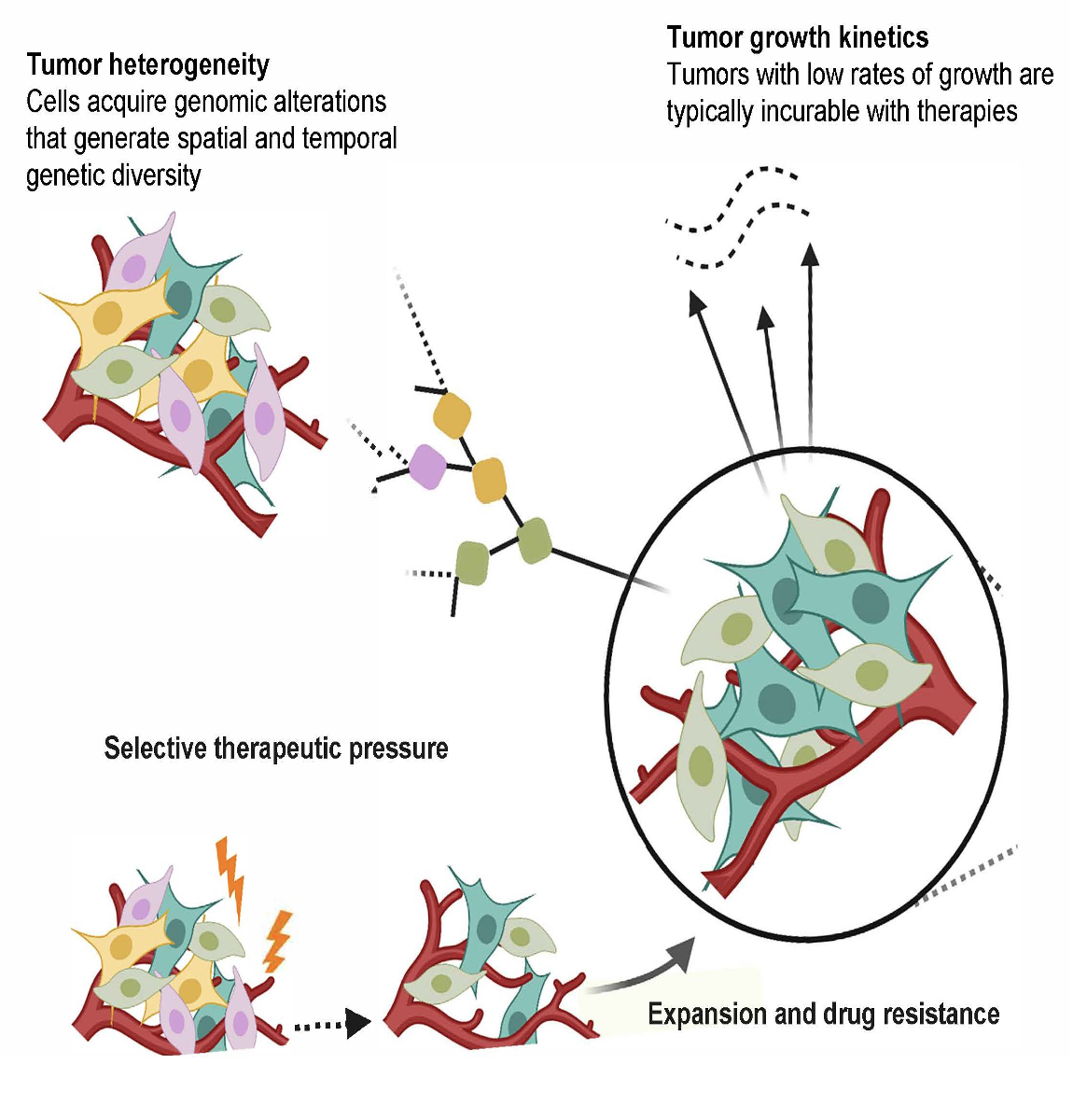
The overall goal of Project 3 is the organoid-based definition of mechanisms of acquired resistance (AR) to targeted therapy in non-small cell lung cancer (NSCLC).
Aim 1:
Determination of synthetic lethal interactions that overcome acquired resistance to osimertinib.
Aim 2:
Characterization of progression toward acquired drug resistance in ALI organoid cultures.
Aim 3:
Investigation of TME effects on acquired resistance to mutant EGFR and KRAS inhibitors.
(For additional information, please visit NIH RePORTER)